(presented 1702, published 1743) "Discours sur quelques propriétés de l'Air, & le moyen d'en connoître la température dans tous les climats de la Terre" (Discourse on some properties of air and on the means of knowing the temperature in all climates of the Earth), Mémoires de l’Académie des sciences de Paris, 155–74. (presented 1699, published 1732) "Moyens de substituer commodément l'action du feu à la force des hommes et des chevaux pour mouvoir les machines" (Ways to conveniently substitute the action of fire for the force of men and horses to power machines), Mémoires de l’Académie des sciences de Paris (presented 1699, published 1732), 112–26 see especially pp. Charles avait remarqué depuis 15 ans la même propriété dans ces gaz mais n'avant jamais publié ses résultats, c'est par le plus grand hasard que je les ai connus." (Before going further, I should inform that although I had recognized many times that the gases oxygen, nitrogen, hydrogen, and carbonic acid, and atmospheric air also expand from 0° to 80°, citizen Charles had noticed 15 years ago the same property in these gases but having never published his results, it is by the merest chance that I knew of them.) On page 157, Gay-Lussac mentions the unpublished findings of Charles: " Avant d'aller plus loin, je dois prévenir que quoique j'eusse reconnu un grand nombre de fois que les gaz oxigène, azote, hydrogène et acide carbonique, et l'air atmosphérique se dilatent également depuis 0° jusqu'a 80°, le cit. (1802), "Recherches sur la dilatation des gaz et des vapeurs", Annales de Chimie, 43: 137–75. "On the expansion of elastic fluids by heat," Memoirs of the Literary and Philosophical Society of Manchester, vol. On the force of steam or vapour from water and various other liquids, both in vacuum and in air" and Essay IV. Thermal expansion – Tendency of matter to change volume in response to a change in temperature.Hand boiler – glass sculpture sometimes used as a collector's item to measure love Pages displaying wikidata descriptions as a fallback.Ideal gas law – Equation of the state of a hypothetical ideal gas.Avogadro's law – Relationship between volume and amount of a gas at constant temperature and pressure Charles law states for a fixed amount of an ideal gas its volume is directly proportional to its temperature at constant pressure.Gay-Lussac's law – Relationship between pressure and temperature of a gas at constant volume.Combined gas law – Combination of Charles', Boyle's and Gay-Lussac's gas laws.Boyle's law – Relationship between pressure and volume in a gas at constant temperature.If you appreciate our work, consider supporting us on ❤️ patreon.V T = k, or V = k T See also Volume vs temperature (in ☌) at different pressures Associated articles The same is true for the below figure except each of the lines converges at −273.15 ☌. 
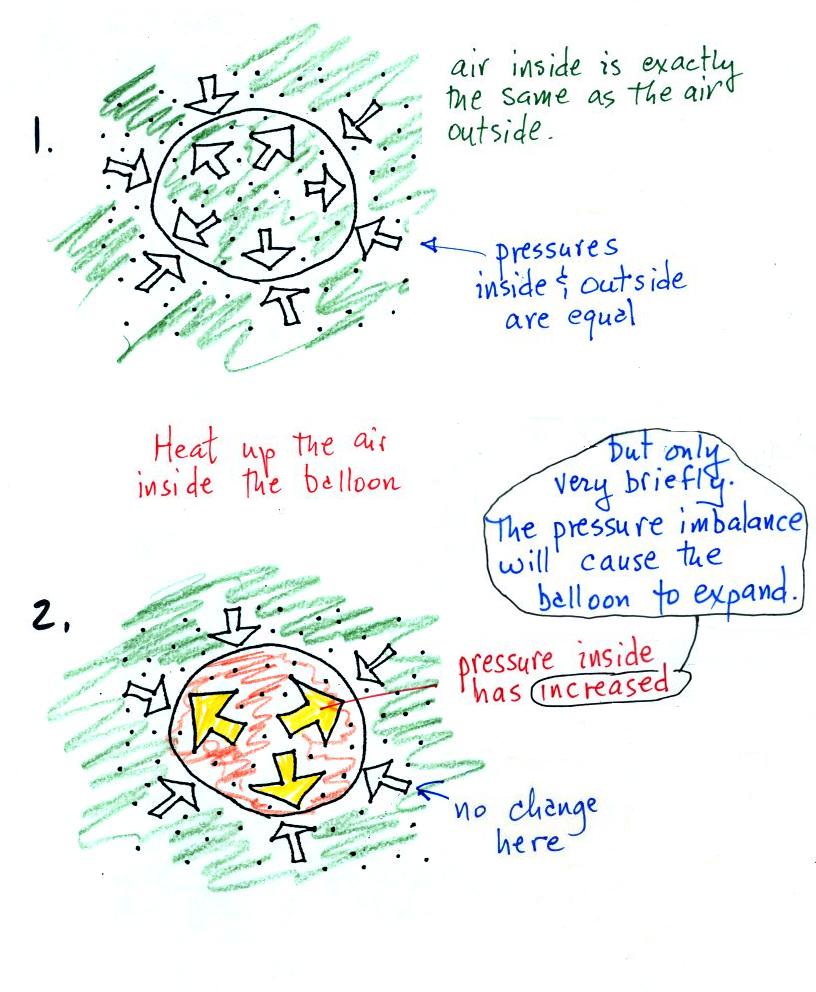
This is because of a decrease in the value of k with an increase in pressure. Also, if noticed, with an increase pressure, the lines shift inwards (towards x-axis). Volume vs temperature (in K) at different pressuresĪs we can see in the above figure, each of the lines converges at zero volume as temperature tends to zero. Such lines that are drawn at a constant pressure are called isobars. Each of the lines is at a constant pressure. The graphs below show four different lines. The above plots can be plotted at different pressures. This is also the equation of Charles's law when temperature is expressed in the degree celsius.Īs from the above figure, when temperature approaches −273.15 ☌, volume also approaches zero. Here, V 0 is the volume at the freezing point of water, t is temperature in the degree celsius, and V is volume at temperature t. Charles law at constant pressure, PV nRT here, n and R so, V T V 1 T 1 V 2 T 2 are constants. The equation of the line in the above figure is. Charles Law is the law in which the temperature is directly proportional to the volume of the container at constant pressure. The plot is a straight line making x-intercept at −-273.15 ☌ I think that Charles' Law states that varying the temperature causes a proportional change in the volume, not the other way around. However, we can also plot the graph of Charles's law with temperature in the celsius scale. In the above plot, the temperature axis is in the kelvin scale (absolute scale). Charles's Law states that the volume of a given mass of gas varies directly with the absolute temperature of the gas when pressure is kept constant. The graph of Charles's law in the celsius scale Experiment 4 Charles’ Law 42 Experiment 4 Charles’ Law The most common statement of Charles’ Law is The volume of a fixed quantity of gas at constant pressure varies linearly with its absolute (Kelvin) temperature. Charles's Law French physicist Jacques Charles (1746-1823) studied the effect of temperature on the volume of a gas at constant pressure. As temperature approaches absolute zero (0 K), the real gases start deviating significantly from Charles's law. 
Note: Real gases do not obey Charles's law at low temperatures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
